Response Surface Methodology–Based Optimization of HPLC Conditions for Quantification of Paracetamol in Indonesian Traditional Medicines (Jamu)
DOI:
https://doi.org/10.22452/mjs.vol45no1.5Keywords:
Jamu for hyperuricemia, Paracetamol, HPLC, Response Surface Methodology, Box-Behnken DesignAbstract
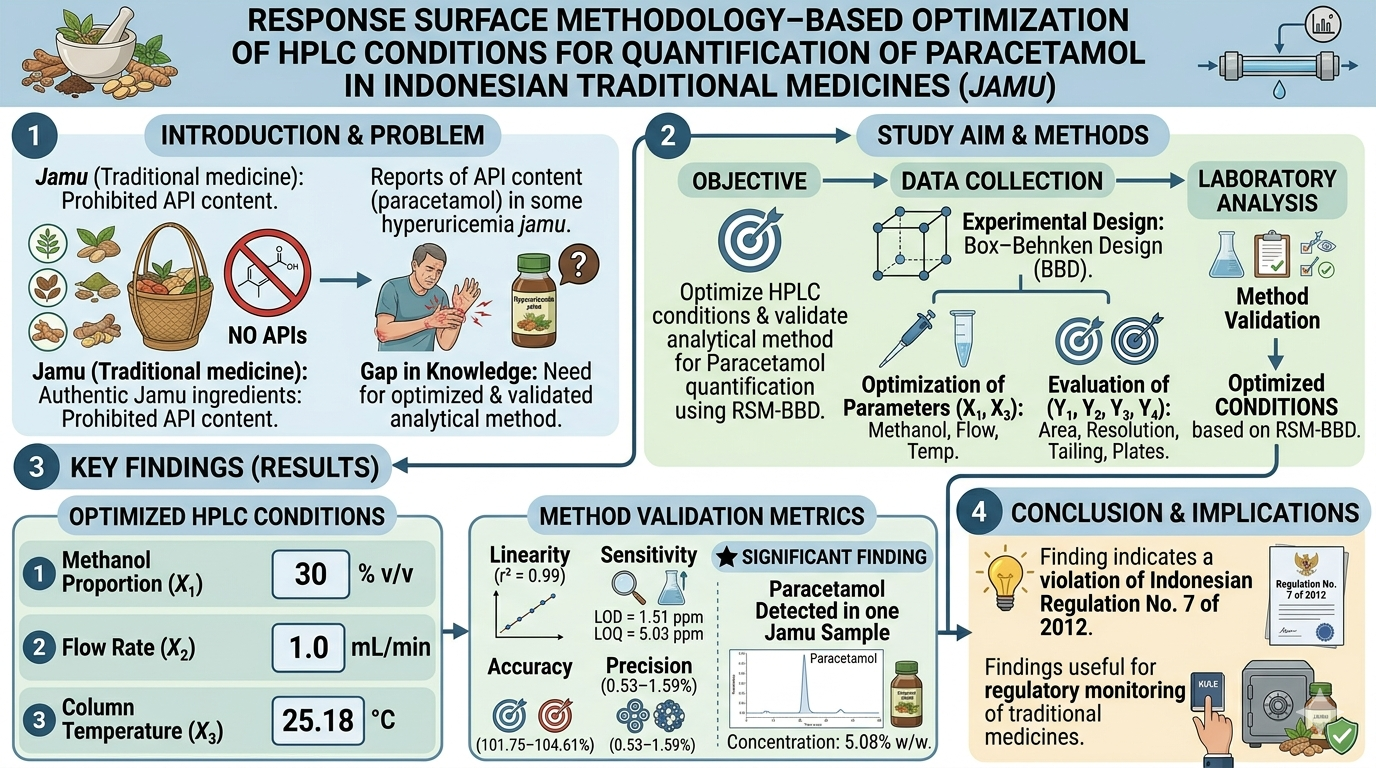
Indonesian traditional herbal medicines (Jamu) are prohibited from containing active pharmaceutical ingredients (APIs). Nevertheless, reports indicate that some hyperuricemia jamu products still contain such substances, including paracetamol This study aimed to optimize High-Performance Liquid Chromatography (HPLC) conditions and to validate an analytical method for the determination of paracetamol in jamu for hyperuricemia obtained from Pasar Besar, Malang City. Response Surface Methodology (RSM) combined with a Box–Behnken Design (BBD) was employed to optimize the HPLC conditions. The investigated parameters were the methanol proportion in the mobile phase (X₁, 30–90% v/v), flow rate (X₂, 0.5–1.0 mL/min), and column temperature (X₃, 20–30 °C). The evaluated response variables included peak area (Y₁), resolution (Y₂), tailing factor (Y₃), and theoretical plate number (Y₄). The optimal HPLC conditions consisted of 30% v/v methanol in aquadest, a flow rate of 1.0 mL/min, and a column temperature of 25.18 °C. Method validation demonstrated satisfactory selectivity (λmax 245 nm; paracetamol retention time ± 3.33 min), linearity (r² = 0.99), limit of detection (LOD) of 1.51 ppm, and limit of quantification (LOQ) of 5.03 ppm. The method also showed acceptable accuracy (101.75–104.61%) and precision (0.53–1.59%), fulfilling the acceptance criteria. Paracetamol was detected in one jamu sample for hyperuricemia at an average concentration of 5.08% w/w. This finding indicates a violation of the Regulation of the Minister of Health of the Republic of Indonesia No. 7 of 2012 concerning the registration of traditional medicines.
References
AOAC International. (2013). Guideline for Dietary Supplements and Botanical (Appendix K). AOAC Off. Method Anal, 8–12.
Ashraful Islam, S., Md Abuzar, S., & Kumar Paul, P. (2011). Validation of UV-Spectrophotometric and RP-HPLC methods for the simultaneous analysis of Paracetamol and Aceclofenac in marketed tablets. Int. J. of Pharm. & Life Sci. (IJPLS), 2(12), 1267–1275.
Azmi, S. N. H., Al Hoqani, U., Al Mamari, J. O. S., Al Mamari, B. M. S., Al Jassasi, B. S. A. R., Al Rubaiai, A. S. S., Rahman, N., Nasir, M., Haque, S. M., Khan, S. A., Ahmed, Q. U., & Zakaria, Z. A. (2022). Box–Behnken Design Based Development of UV-Reversed Phase High Performance Liquid Chromatographic Method for Determination of Ascorbic Acid in Tablet Formulations. Separations, 9(11), 1–21. https://doi.org/10.3390/separations9110361
Chidiac, A. S., Buckley, N. A., Noghrehchi, F., & Cairns, R. (2023). Paracetamol (acetaminophen) overdose and hepatotoxicity: mechanism, treatment, prevention measures, and estimates of burden of disease. Expert Opinion on Drug Metabolism and Toxicology, 19(5), 297–317. https://doi.org/10.1080/17425255.2023.2223959
Fitrianasari, R. D. P., Haresmita, P. P., & Agusta, H. F. (2023). Qualitative Analysis of Drug Substances in Rheumatic Jamu Samples Using Thin Layer Chromatography. Medical Sains : Jurnal Ilmiah Kefarmasian, 8(3), 999–1008. https://doi.org/10.37874/ms.v8i3.862
Husain, F., Ysrafil, Y., Daud, R. P. A., Aisyah, A. N., Fadri, A., & Nur, S. (2023). Authentication of Medicinal Chemicals in Traditional Herbal Products (Jamu) by UV-Vis Spectrophotometry. Hacettepe University Journal of the Faculty of Pharmacy, 43(2), 100–110. https://doi.org/10.52794/hujpharm.1090673
Ihsan, B. R. P., Nurhayati, I. P., & Maysaroh, I. (2022). Performance Chromatography Double Mass Spectrometry ( UHPLC-MS / MS ) Validasi Metode Ultra High Performance Chromatography Double Mass Spectrometry ( UHPLC-MS / MS ) untuk Analisis Kurkumin pada Ekstrak Etanol Kunyit ( Curcuma longa ) dengan Berbagai Per. Pharmaceutical Journal of Indonesia, 4(1), 29–34. https://doi.org/10.21776/ub/pji
Kementerian Kesehatan Republik Indonesia. (2020). Farmakope Indonesia edisi VI. In Departemen Kesehatan Republik Indonesia.
McCrae, J. C., Morrison, E. E., MacIntyre, I. M., Dear, J. W., & Webb, D. J. (2018). Long-term adverse effects of paracetamol – a review. British Journal of Clinical Pharmacology, 84(10), 2218–2230. https://doi.org/10.1111/bcp.13656
Pratama, M., Harisman, D., Labasy, L., Seniwati, & Fawwaz, M. (2022). Determination of Acetaminophen in Jamu Pegal Linu by High Performance Liquid Chromatography. Research Artikel, Iii, 12–15.
Putri, N., Sari, D., & Haresmita, P. P. (2023). PEGAL LINU DI WILAYAH MAGELANG. Jurnal Ilmu Farmasi Dan Farmasi Klinik (JIFFK), 20(1), 53–59.
Saragiotto, B. T., Abdel Shaheed, C., & Maher, C. G. (2019). Paracetamol for pain in adults. The BMJ, 367(December), 1–5. https://doi.org/10.1136/bmj.l6693
Savitri, D. (2017). Diam-diam Mematikan, Cegah Asam Urat dan Hipertensi. Penerbit Anak Hebat Indonesia.
Sumarni, W., Sudarmin, S., & Sumarti, S. S. (2019). The scientification of jamu: A study of Indonesian’s traditional medicine. Journal of Physics: Conference Series, 1321(3). https://doi.org/10.1088/1742-6596/1321/3/032057
Taupik, M., Djuwarno, E. N., Mustapa, M. A., Kunusa, W. R., La Kilo, J., & Sahumena, M. H. (2022). The Type Fragmentation Patterns Confirmed Acetaminophen By Using Liquid Chromatography-Mass Spectroscopy (LCMS) From Herbal Medicine (Jamu). Elkawnie: Journal of Islamic Science and Technology, 7(2), 341. https://doi.org/10.22373/ekw.v7i2.7492
Wisnuwardhani, H. A., Rusdi, B., & Yuliawati, K. M. (2018). Method validation for simultaneous quantitative analysis of acetaminophen and dexamethasone in jamu pegal linu using spe-hplc method. Journal of Pharmaceutical Sciences and Research, 10(11), 2693–2696.
Downloads
Published
Issue
Section
License
Copyright (c) 2026 Malaysian Journal of Science (MJS)

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
Transfer of Copyrights
- In the event of publication of the manuscript entitled [INSERT MANUSCRIPT TITLE AND REF NO.] in the Malaysian Journal of Science, I hereby transfer copyrights of the manuscript title, abstract and contents to the Malaysian Journal of Science and the Faculty of Science, University of Malaya (as the publisher) for the full legal term of copyright and any renewals thereof throughout the world in any format, and any media for communication.
Conditions of Publication
- I hereby state that this manuscript to be published is an original work, unpublished in any form prior and I have obtained the necessary permission for the reproduction (or am the owner) of any images, illustrations, tables, charts, figures, maps, photographs and other visual materials of whom the copyrights is owned by a third party.
- This manuscript contains no statements that are contradictory to the relevant local and international laws or that infringes on the rights of others.
- I agree to indemnify the Malaysian Journal of Science and the Faculty of Science, University of Malaya (as the publisher) in the event of any claims that arise in regards to the above conditions and assume full liability on the published manuscript.
Reviewer’s Responsibilities
- Reviewers must treat the manuscripts received for reviewing process as confidential. It must not be shown or discussed with others without the authorization from the editor of MJS.
- Reviewers assigned must not have conflicts of interest with respect to the original work, the authors of the article or the research funding.
- Reviewers should judge or evaluate the manuscripts objective as possible. The feedback from the reviewers should be express clearly with supporting arguments.
- If the assigned reviewer considers themselves not able to complete the review of the manuscript, they must communicate with the editor, so that the manuscript could be sent to another suitable reviewer.
Copyright: Rights of the Author(s)
- Effective 2007, it will become the policy of the Malaysian Journal of Science (published by the Faculty of Science, University of Malaya) to obtain copyrights of all manuscripts published. This is to facilitate:
- Protection against copyright infringement of the manuscript through copyright breaches or piracy.
- Timely handling of reproduction requests from authorized third parties that are addressed directly to the Faculty of Science, University of Malaya.
- As the author, you may publish the fore-mentioned manuscript, whole or any part thereof, provided acknowledgement regarding copyright notice and reference to first publication in the Malaysian Journal of Science and Faculty of Science, University of Malaya (as the publishers) are given. You may produce copies of your manuscript, whole or any part thereof, for teaching purposes or to be provided, on individual basis, to fellow researchers.
- You may include the fore-mentioned manuscript, whole or any part thereof, electronically on a secure network at your affiliated institution, provided acknowledgement regarding copyright notice and reference to first publication in the Malaysian Journal of Science and Faculty of Science, University of Malaya (as the publishers) are given.
- You may include the fore-mentioned manuscript, whole or any part thereof, on the World Wide Web, provided acknowledgement regarding copyright notice and reference to first publication in the Malaysian Journal of Science and Faculty of Science, University of Malaya (as the publishers) are given.
- In the event that your manuscript, whole or any part thereof, has been requested to be reproduced, for any purpose or in any form approved by the Malaysian Journal of Science and Faculty of Science, University of Malaya (as the publishers), you will be informed. It is requested that any changes to your contact details (especially e-mail addresses) are made known.
Copyright: Role and responsibility of the Author(s)
- In the event of the manuscript to be published in the Malaysian Journal of Science contains materials copyrighted to others prior, it is the responsibility of current author(s) to obtain written permission from the copyright owner or owners.
- This written permission should be submitted with the proof-copy of the manuscript to be published in the Malaysian Journal of Science
Licensing Policy
Malaysian Journal of Science is an open-access journal that follows the Creative Commons Attribution-Non-commercial 4.0 International License (CC BY-NC 4.0)
CC BY – NC 4.0: Under this licence, the reusers to distribute, remix, alter, and build upon the content in any media or format for non-commercial purposes only, as long as proper acknowledgement is given to the authors of the original work. Please take the time to read the whole licence agreement (https://creativecommons.org/licenses/by-nc/4.0/legalcode ).





